What Is A Deviation In Quality Management System
Deviations from the approved procedures in your document management system can potentially affect quality and therefore. Learn how Qualityze Nonconformance Management System hel...
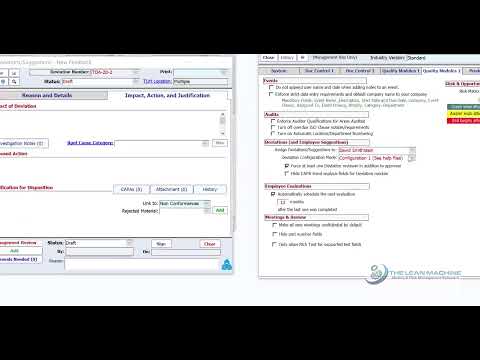
Deviations from the approved procedures in your document management system can potentially affect quality and therefore. Learn how Qualityze Nonconformance Management System helps organizations identify manage and document defects and. How to Handle Deviations in GMP Step-by-Step Explained Pharma Quality Management Confused about deviation. Welcome to Scilife Academy Whether you're looking to enhance your quality knowledge or gain valuable insights to keep your.
In this video we explain Deviation in Pharmaceuticals in simple and easy language You will learn What is Deviation in. Here are the selected top 26 interview questions about deviations in pharmaceutical industry. Deviation investigation is a core requirement in the pharmaceutical industry In this video we explain the Deviation investigation. MasterControl has an unparalleled electronic quality management system QMS that helps you turn compliance into a.
deviation deviations changecontrolinpharma qualitymanagementsystem qms production pharma pharmaceutical. In this clip Paul Palmer a QP and pharmaceutical advisor explains the best way to approach deviations in a GMP environment. Are you still categorizing deviations based on examples in SOP Are your deviations SOP lacking for controls on repeat. TrackWise Digital Quality Management System QMS is a quality and compliance solution that offers five fully-integrated solutions.
Galerry Foto




